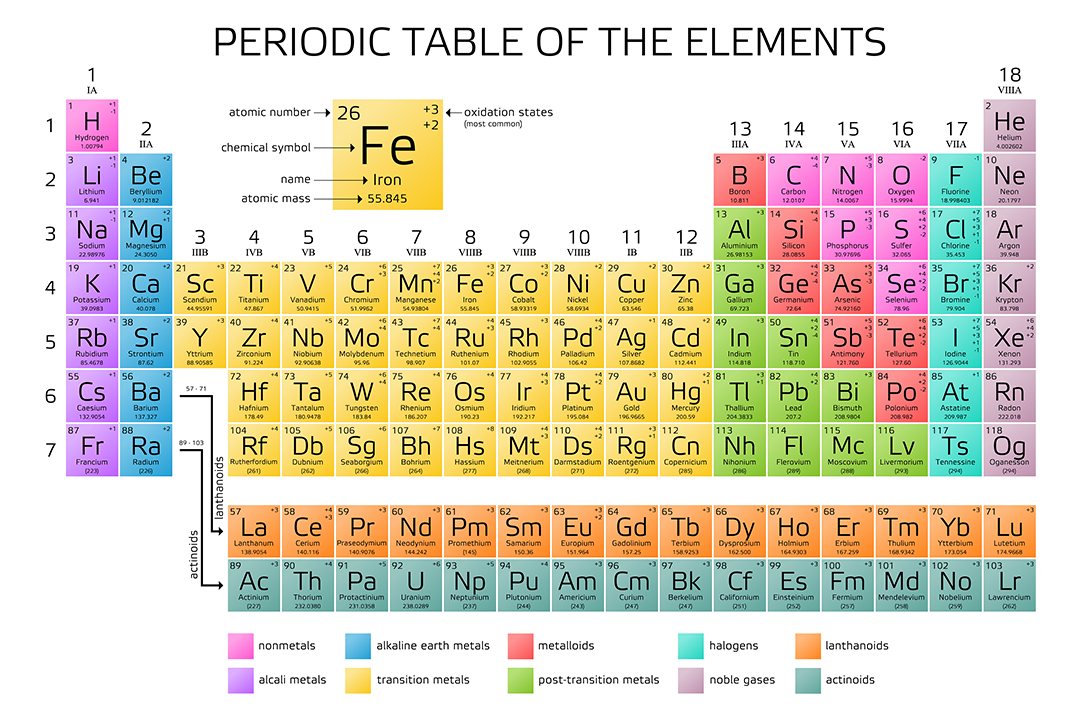 
Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole. The division sign (/) implies “per,” and “1” is implied in the denominator. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). Interactive periodic table with up-to-date element property data collected from authoritative sources. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. adenosine (C 10H 13N 5O 4), a component of cell nuclei crucial for cell divisionīe careful when counting atoms.Find out the meanings of the periods, groups, and symbols of the periodic table, and how to work with the atomic weights and molar weights of the elements. This table can be obtained in PDF format here. Learn how to use the periodic table to convert between molar masses and moles of any element, and how to use the relative molar masses of the elements to calculate their molar masses. The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. From the periodic table, the atomic mass of hydrogen is 1.008 g/mol, and the atomic mass of. Prepare a concept map and use the proper conversion factor. This black and white periodic table contains the accepted atomic weights of each element as accepted by the IUPAC. Simply calculating the sum of the atomic mass of 2 H and 1 O will provide the molar mass of water. barium sulfate (BaSO 4), used to take X rays of the gastrointestional tract This periodic table contains the atomic masses of the elements as accepted by the IUPAC (09-2013).3: Table salt, NaCl, contains an array of sodium and chloride ions combined in a 1:1 ratio. The formula mass for this compound is computed as 58.44 amu (Figure 3.6.3 3.6. What is the mass of 1 mol of each substance? Sodium chloride is an ionic compound composed of sodium cations, Na +, and chloride anions, Cl, combined in a 1:1 ratio. You will need to refer to a periodic table for proton values.\): Moles to Mass Conversion with Compounds In the periodic table, the vertical columns are called groups and the horizontal rows are called periods. In this notation, the atomic number is not included. Periodic Table of Elements - The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. Symbol-mass format for the above atom would be written as Cr-52. So, Molar mass of O2 Molar mass of 2 Oxygen (O) atoms. The atomic masses of some elements are known very precisely, to a large number of decimal places. You can see that in O2, there are 2 Oxygen atoms. The atomic masses in Table A.1 The Basics of the Elements of the Periodic Table represent the number of decimal places recognized by the International Union of Pure and Applied Chemistry, the worldwide body that develops standards for chemistry. You can see that The molar mass of Potassium is 39.098 g/mol. So let’s look at the molar mass of Potassium and Bromine from the above periodic table. Now in KBr, there is 1 Potassium atom and 1 Bromine atom. 1 Now, to calculate the molar mass of O2, you just have to add the molar mass of all the individual atoms that are present in O2. You can see the molar mass value of all the atoms from this periodic table. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The molar mass of Oxygen is 15.999 g/mol. So for that, have a look at the periodic table given below. Now here we have to find the molar mass of FeCl3. Because the molar mass of any molecule (or compound) can be calculated by simply adding the molar masses of individual atoms. The "A" value is written as a superscript while the "Z" value is written as a subscript. If you have a periodic table with you, then you can easily calculate the molar mass of FeCl3. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
